Serviços Personalizados
Artigo
Links relacionados
Compartilhar
RFO UPF
versão impressa ISSN 1413-4012
RFO UPF vol.20 no.1 Passo Fundo Jan./Abr. 2015
Evaluation of three instrumentation techniques with different apical diameters on smear layer formation
Avaliação de três técnicas de instrumentação com diferentes diâmetros apicais na formação de smear layer
Dilene PorciúnculaI; Rogério LossI; Fernanda Ullmann LopezII; Leticia Boldrin MestieriIII; Fabiana Soares GreccaIV; Marcus Vinícius Reis SóIV
I DDS, Dentist, Clinical Department, School of Dentistry, Cruzeiro do Sul University (UNICSUL), Caxias do Sul, RS, Brazil
II DDS, MSc, PhD student, Clinical Department, School of Dentistry, Pontifical Catholic University of Rio Grande do Sul - PUCRS, Porto Alegre, RS, Brazil
III DDS, MSc, PhD student, Department of Conservative Dentistry, School of Dentistry, Federal University of Rio Grande do Sul - UFRGS, Porto Alegre, RS, Brazil
IV DDS, MSc, PhD, Professor of Endodontics, Department of Conservative Dentistry, School of Dentistry, Federal University of Rio Grande do Sul - UFRGS, Porto Alegre, RS, Brazil
RESUMO
Objetivo: avaliar os efeitos de diferentes instrumentações na formação de smear layer no terço apical de 90 raízes mesiobucais de molares superiores por microscopia eletrônica de varredura (MEV). Materiais e método: três grupos foram formados baseados nas técnicas utilizadas: instrumentação manual, sistema rotatório K3 e sistema reciprocante NSK. Os grupos foram subdivididos em três, de acordo com o diâmetro apical: #30, #35 ou #40. Após o preparo, as raízes foram seccionadas no sentido transversal, separando os terços apicais do restante das raízes; esses terços foram divididos em duas metades e preparados para MEV. A formação de smear layer na superfície do canal radicular e os túbulos dentinários foram avaliados por escores num aumento de 1.000×. Os dados foram analisados pelo teste Kruskal-Wallis complementado pelo teste de Dunn (p < 0.05). Resultados: o aumento no diâmetro do preparo apical não influenciou na formação de smear layer nas paredes dentinárias. Conclusão: apesar das técnicas e dos diâmetros apicais utilizados durante o preparo, nenhuma parede livre de smear layer foi observada.
Palavras-chave: Endodontia. Preparo do canal radicular. Smear layer.
ABSTRACT
Objective: to evaluate the effects of different instrumentations on smear layer formation in the apical third of 90 mesio-buccal roots of upper molars by scanning electron microscopy (SEM). Materials and method: three groups were formed based on techniques used: hand instrumentation, K3 rotary system, and NSK reciprocating handpiece. The groups were subdivided by three according to the size of the apical diameter: #30, #35 or #40 files. After preparation, roots were transversely sectioned, separating the apical thirds of the remaining roots; these thirds were split into halves and prepared for SEM. The smear layer formation on the surface of the root canal and in dentinal tubules was scored at 1,000× magnification. Data were analyzed using the Kruskal– Wallis test and Dunn's test (p < 0.05). Results: the increase of the diameter in apical preparation had no influence on smear layer formation on dentinal walls. Conclusion: regardless of the technique and apical diameters applied for preparation, no dentinal wall free of smear layer was observed.
Keywords: Endodontics. Root canal preparation. Smear layer.
Introdução
The mechanical preparation of root canals plays a significant role in endodontic therapy. The process involves the removal of pulp cavity contents and the consequent reduction of microorganism counts, especially in non-vital teeth with periapical lesions. On the other hand, root canal instrumentation produces a smear layer on dentinal walls where the endodontic instrument works, containing dentin debris, organic remains such as pulp tissue, odontoblast processes, necrotic debris, and microorganisms and their metabolic products1-2.
Literature affirms that canal anatomy plays a more important role in endodontic treatment. Isthmuses, curvatures, and oval-shaped canals make disinfection of the root canal system challenging3. Previous studies indicated that no existing instrumentation techniques are sufficient for completely cleaning canal walls4-6.
There are contradictory findings in literature regarding as to whether apical enlargement is necessary7- 9. Regarding the importance of the canal taper in curved canals, some authors observed that the total removal of the smear layer occurred only in preparations with an apical diameter of at least 0.30 mm10.
In this context, this study evaluated the effect of three different root canal preparation techniques with three different apical diameters on smear layer formation in the apical third of upper mesio-buccal roots, by SEM.
Materials and method
This study was approved by the Research Ethics Committee at the Federal University of Rio Grande do Sul, Brazil (N. 196/06).
Ninety extracted human maxillary molars were radiographed in the bucco-lingual and mesio-distal direction. Teeth were excluded from the study if they showed previous endodontic manipulation, incomplete root formation, calcification, internal or external resorption, or curvature greater than 40°.
Dental crowns were sectioned below the cementum- enamel junction and the mesio-buccal root was separated from the other roots, so that every root was 12-13 mm long. Canals were irrigated with 1% sodium hypochlorite (NaOCl; Farmácia Marcela, Porto Alegre, RS, Brazil) and accessed by a size 10 K file (Denstply Maillefer, Ballaigues, Switzerland). The working length was calculated by subtracting 1 mm from the point at which the file tip protruded from the apical foramen.
A size 10 file was inserted up to the working length and fixed with wax. Thereafter, a radiograph was obtained (Dabi Atlante Spectro, 70 KVp, Ribeirão Preto, SP, Brazil) and the images of each canal were scanned and transferred to Auto Cad 2000 software (Autodesk Incorporated, San Rafael, CA, USA).
Root canal curvatures were mathematically determined by means of measurement of angles and radii11,12. The mean curvature angle was 15°-40°. An equal number of roots with similar degrees of curvature and radii were randomly distributed into three groups with 30 samples each.
Each group was then subdivided into three groups (n=10) where the apical preparation was carried out with size 30, 35, and 40 files.
Group 1: Thirty roots were prepared crown-down using Flexofile manual files (Denstply Maillefer Ballaigues, Switzerland), starting with a size 45 or 40 file and using balanced force movements. This step was repeated with instruments of successively smaller sizes until the working length was reached. Files used in the apical third of the root canal had their last few millimeters pre-curved by an endodontic file bender (Flexobend, Aronson, SP, Brazil). After apical stop preparation (#30, #35 or #40), a step-back procedure was performed using four instruments with sizes greater than the final apical file with anti-curvature filing motions.
Group 2: Thirty roots were instrumented using the K3 system (SybronEndo, Glendora, CA, USA) at 300 rpm with torque set at 2 N. The cervical third was prepared using orifice shapers #25/.10 and #25/.08, the mid-third was prepared with #30/.06 and #25/.04 files, and the apical third was prepared using #30/.02, #35/.02, or #40/.02 files, according to each group.
Group 3: Thirty roots were instrumented using a NSK reciprocating handpiece and Flexofile stainless steel manual files. The technique used introduced the greatest file size (size 45, 40) in the cervical third until achieving traction and resistance to anti-curvature filing motions. Progressively smaller instruments were then used until the working length was reached. Files used in the apical third of the root canals had their last few millimeters pre-curved. After apical stop preparation (#30, #35 or #40) a step-back procedure was performed (i.e., four instruments with sizes greater than the final apical file).
After each instrument change, canals were irrigated with 2.0 mL of 1% NaOCl, delivered by 10-mL disposable syringes and 30-gauge needles (Navitip, Ultradent, South Jordan, USA), and placed to approximately 3-4 mm from the working length without binding. Recapitulation was performed with a size 10 stainless steel file up to the working length. Upon completion of instrumentation a final irrigation was performed with 10-mL of 17% trisodium ethylenediaminetetraacetic acid (EDTA; Farmácia Marcela, Porto Alegre, RS, Brazil) for 3 minutes; next the root canal was immediately washed with 2.0 mL of 1% NaOCl solution followed by copious rinsing with 4.0 mL saline solution.
Each instrument was used to prepare five root canals and subsequently discarded. The canals were then dried using paper points (Tanari Industrial Ltda., Manaus, AM, Brazil). A small cotton pellet was placed inside the coronal opening of each canal to prevent dentinal shavings or debris from entering the root canal during sectioning.
Finally, two longitudinal grooves were prepared on the buccal and lingual surfaces of each root using a diamond disc without penetrating into the canal. The roots were then split into halves with a chisel. The apical third was separated from the remaining roots, and were conserved and coded. The coded specimens were then mounted on metallic stubs, gold sputtered, and examined by SEM (JEOL 6060, Tokyo, Japan) at 1,000 × magnification.
The images were analyzed and scored in a computer monitor. The amount of smear layer remaining on the surface of the apical root canal or in dentinal tubules was scored according to the following criteria13: 1- no smear layer: no smear layer detected on the surface of root canals, and all tubules clean and open; 2- moderate smear layer: no smear layer is observed on the surface of root canals, but tubules contain debris; and 3- heavy smear layer: the smear layer covers the root canal surface and tubules. An endodontist was blinded to the coding system so to exclude observer bias, and calibrated according to ICC (Intraclass Correlation).
Data were analyzed using the Kruskal–Wallis and Dunn's tests. The level of significance was set at 5%.
Results
Intra-observer calibration showed an agreement ratio of 94%.
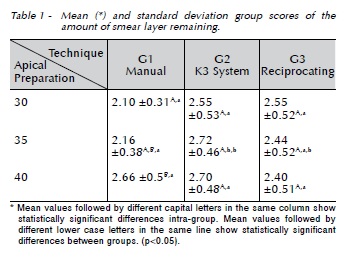
The smear layer scores in each group are presented in Table 1 and Figure 1. It was found that within the same group with varying apical diameters only the manual technique showed differences for sizes 30 and 40, wherein a higher amount of smear layer was observed when the #40 file was used (p < 0.05)..When the techniques were compared to each instrument, only size 35 resulted in a statistically significant difference (p = 0.028).

Discussion
This research was designed to answer a question that continues to cause disagreement and is widely discussed in endodontic studies: widening of curved and flattened canals, such as those of the mesial roots of upper and lower molars with pulp
necrosis and apical lesions, by using size 35 and 40 tapered files for apical preparation14.
Several researches regarding debris and smear layer have shown that neither instruments nor instrumentation techniques are capable of achieving complete cleanliness of root canal walls4. Whereas most of the debris was removed, different degrees of smear layer were found in all root canals5,15-17.
Debris may be defined as dentine chips, tissue remnants, and particles loosely attached to the root canal wall18, whilst the smear layer is a surface film of debris retained on dentine or other surfaces after instrumentation with either rotary instruments or endodontic files19.
The results of this study also pointed that regardless of the technique and the diameter of the apical instrument applied for preparation, dentinal walls (i.e., open dentinal tubules and sharp outlines on the level of the apical third) free of smear layer were not observed. It is important to note that any previous studies that took advantage of the increased diameter of apical preparation and sought to determine the association between the amount of the remaining smear layer using SEM were not found.
In all three groups, the smear layer score was between 2 and 3, representing the smear that partially blocked dentinal tubules. Furthermore, group 1, size 35 showed better effectiveness for cleaning than did the continuously rotating one. This finding corroborate the reports from other authors that most cases of manual instrumentation proved to be superior to mechanical instrumentation as far as cleaning effectiveness is concerned20. (Manjunatha et al., 2013).
Although the statistical analysis point to a lower amount of smear layer for file #30 compared with #40 in manual instrumentation, this seems to be clinically irrelevant, since the scores were above 2, which characterizes the presence of dentinal tubules blocked by the smear layer. It is speculated that, in group 1, when #30 and #35 files were used in the apical third, softer and rhythmic instrumentation is required with less risk of diversion and compaction of dentine than when a #40 file is used.
To evaluate smear layer formation, this study was based on the cleaning score previously established13,21. The choice of the apical third for evaluation was appropriate for achieving the results, considering this region is critical to achieve shaping and cleaning. Moreover, the apical third of the mesio-buccal root presents a circular cross-section, allowing for better instrument action working on dentinal walls2, and so justifying the scores.
An evaluation of cervical and middle third could mask results because at this level the MB canal has a flat cross-section or even comma-shaped, so that the instruments leave untouched areas on dentinal wall, allowing to observe open dentinal tubules and pulp tissue remnants1,22. The effects of root canal anatomy on the morphology after preparation were analyzed by computed microtomography and reported that NiTi rotary instruments left nearly 35% of untouched dentinal surface23.
The evaluation of preparation taper using size 20 or size 40 ProFile GT files, and the ability to introduce irrigant and remove debris from root canals suggest that debris is more effectively removed when the apical preparation size is larger (size 40) than size 20.
However, these authors compared #20 and #40 files with different tapers and performed the analysis for debris15. One source of bias in studies of this kind is the selection of teeth20. The authors stated that identical shapes of root canals in natural teeth are almost impossible to obtain. However, it is essential to use natural teeth in studies such as these.
Teeth with apical lesions have a greater bacterial propagation in dentin tubules, so a larger apical size preparation is required for better disinfection24. In addition, literature has shown that a minimum apical size preparation #30 is needed for the irrigant to penetrate in the apical third of the root canal15.
This study did not evaluate the cleaning capacity of irrigating solutions. Nonetheless, there is evidence that the flushing action of the irrigant solution and the ability to dissolve tissue are important during the cleaning process25.
Concerning the shaping abilities of manual and mechanical instrumentation techniques investigated in this research, a comparison study including instrumentation results both in narrow and curved root canal has been previously published14. Best results were obtained for the continuous rotation with less apical transportation when apical preparation was carried out with greater sizes like #35 and #40 taper 0.02 mm/mm.
In the light of these issues, it becomes clear that larger apical preparation in infected root canals is required. In cases involving curved and flattened canals nickel-titanium rotary instrumentation seems to be the most effective and safe method for preparation. So a better technique for removing the smear layer produced by larger instruments and rotary files must be used. On the other hand, literature brings alternative irrigation systems to increase smear layer removal, which could be improved by apical enlargement. The clinical significance of this smear seems to be clear19. Thus, its presence along with the complex anatomical observations and the presence of microorganisms seem to continue to pose a challenge to clinicians.
Conclusão
It may be reasonably concluded that smear layer is formed, regardless of the technique of canal preparation used. Furthermore, increasing the diameter of the apical preparation does not result in better cleaning of the dentinal walls.
Referências
1. De-Deus G, Reis C. Critical appraisal of published smear layer-removal studies: methodological issues. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2011;112:531-43. [ Links ]
2. Tunga U, Parlak E, Bodrumlu E, Aydemir H, Yesilsoy C. Effect of F-File on removal of the smear layer: A scanning electron microscope study. Aust Endod J. 2011;37:65-9.
3. Adcock JM, Sidow SJ, Looney SW, Liu Y, McNally K, Lindsey R et al. Histologic evaluation of canal and isthmus debridement efficacies of two different irrigant delivery techniques in a closed system. J Endod. 2011;37:544-8.
4. Grecca FS, Garcia RB, Bramante CM, Moraes IG, Bernardineli N. A quantitative analysis of rotary, ultrasonic and manual techniques to treat proximally flattened root canals. J Appl Oral Sci. 2007;15:89-93.
5. Narayan GS, Venkatesan SM, Karumaran CS, Indira R, Ramachandran S, Srinivasan MR. A comparative evaluation on the cleaning and shaping ability of three nickel titanium rotary instruments using computerized tomography - An ex vivo study. Cont Clin Dent. 2012;3:151-155.
6. Zmener O, Pamejer CH, Serrano SA, Hernandez SR. Cleaning efficacy using two engine-driven systems versus manual instrumentation in curved root canals: a scanning electron microscopic study. J Endod. 2011;37:1279-82.
7. De-Deus G, Garcia-Filho P. Influence of the NiTi rotary system on the debridement quality of the root canal space. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;108:e71-6.
8. Interliche R, Marchesan MA, Silva SRC, Pécora JD, Silva-Sousa YTC, Sousa-Neto MD. Influence of Hero apical instruments on cleaning ovoid-shaped root canals. Braz oral Res. 2011;25:314-318.
9. Pécora JD, Capelli A, Guerisoli DM, Spanó JC, Estrela C. Influence of cervical preflaring on apical file size determination. Int Endod J. 2005;38:430-5.
10. Khademi A, Yazdizadeh M, Feizianfard M. Determination of the minimum instrumentation size for penetration of irrigants to the apical third of root canal systems. J Endod. 2006;32:417-20.
11. Schaffer E, Diez C, Hoppe W, Tepel J. Roentgenographic investigation of frequency and degree of canal curvatures in human permanent teeth. J Endod. 2002;28:211-15.
12. Schneider SW. A comparison of canal preparations in straight and curved root canals. Oral Surg Oral Med Oral Pathol. 1971;32:271-75.
13. Torabinejad M, Khademi AA, Babagoli J, Cho Y, Johnson WB, Bozhilov K et al. A new solution for the removal of the smear layer. J Endod. 2003;29:170-5.
14. López FU, Fachin EV, Fontanella VRC, Barletta FB, Só MVR, Grecca FS. Apical transportation: a comparative evaluation of three root canal instrumentation techniques with three different apical diameters. J Endod. 2008;34:1545-48.
15. Albrecht LJ, Baumgartner JC, Marshall JG. Evaluation of apical debris removal using various sizes and tapers of Pro- File GT files. J Endod. 2004;30:425-8.
16. Blank-Gonçalves LM, Nabeshima CK, Martins GHR, Machado MEL. Qualitative analysis of the removal of the smear layer in the apical third of curved roots: conventional irrigation versus activation systems. J Endod. 2011;37:1268-71.
17. Zmener O, Pameijer CH, Serrano SA, Palo RM, Iglesias EF. Efficacy of the NaviTip FX irrigation needle in removing post instrumentation canal smear layer and debris in curved root canals. J Endod. 2009;35:1270-3.
18. Hulsmann M, Peters OA, Dummer PMH. Mechanical preparation of root canals: shaping goals, techniques and means. Endod Topics 2005;10:30-75.
19. Violich DR, Chandler NP. The smear layer in endodontics: a review. Int Endod J. 2010;43:2-15.
20. Manjunatha M, Annapurna K, Sudhakar V, Sunil Kumar V, Hiremath VK, Shah A. Smear layer evaluation on root canal preparation with manual and rotary techniques using EDTA as an irrigant: A Scanning Electron Microscopy Study. J Int Oral Health. 2013;5:66-78.
21. Zand V, Lofti M, Rahimi S, Mokhtari H, Kazemi A, Sakhamanesh HV. A comparative scanning electron microscopic investigation of the smear layer after the use of sodium hypochlorite gel and solution forms as root canal irrigants. J Endod. 2010;36:1234-7
22. Paqué F, Balmer M, Attin T, Peters OA. Preparation of oval-shaped root canals in mandibular molars using nickel-titanium rotary instruments: a micro-computed tomography study. J Endod. 2010;36:703-7.
23. Gergi R, Osta N, Bourbouze G, Zgheib C, Arbab-Chirani R, Naaman A. Effects of three nickel titanium instrument systems on root canal geometry assessed by micro-computed tomography. Int Endod J. 2014;9. [Epub ahead of print].
24. Hübscher W, Barbakow F, Peters OA. Root-canal preparation with FlexMaster: canal shapes analysed by micro-computed tomography. Int Endod J. 2003;36:740-7.
25. Mohammadi, Z. Sodium hypochlorite in endodontics: an update review. Int Dent J. 2008;58:329-41.
 Endereço para correspondência:
Endereço para correspondência:
Fabiana Soares Grecca
Av. Ramiro Barcelos, 2492
90035-003
Porto Alegre-RS, Brazil
e-mail: fabiana.grecca@ufrgs.br
Recebido: 16/09/2014
Aceito: 31/03/2015













