Serviços Personalizados
Artigo
Links relacionados
Compartilhar
Brazilian Journal of Oral Sciences
versão On-line ISSN 1677-3225
Braz. J. Oral Sci. vol.14 no.1 Piracicaba Jan./Mar. 2015
ORIGINAL ARTICLE
Chemical analysis and Vickers hardness of orthodontic mini-implants
Christiane Cavalcante Feitoza I; Guilherme José Pimentel Lopes de Oliveira II; Rafael Leonardo Xediek Consani III; Eloisa Marcantonio Boeck I; Karina Eiras Dela Coleta Pizzol I; Nadia Lunardi I
I Centro Universitário de Araraquara – UNIARA, Department of Dentistry, Area of Orthodontics, Araraquara, SP, Brazil
II Universidade Estadual Paulista – UNESP, Araraquara Dental School, Department of Diagnosis and Surgery, Area of Periodontology, Araraquara, SP, Brazil
III Universidade Estadual de Campinas – UNICAMP, Piracicaba Dental School, Department of Oral Rehabilitation, Piracicaba, SP, Brazil
ABSTRACT
Orthodontic mini-implants are used in clinical practice to provide efficient and aesthetically-pleasing anchorage. Aim: To evaluate the hardness (Vickers hardness) and chemical composition of miniimplant titanium alloys from five commercial brands. Methods: Thirty self-drilling mini-implants, six each from the following commercial brands, were used: Neodent (NEO), Morelli (MOR), Sin (SIN), Conexão (CON), and Rocky Mountain (RMO). The hardness and chemical composition of the titanium alloys were performed by the Vickers hardness test and energy dispersive X-ray spectroscopy, respectively. Results: Vickers hardness was significantly higher in SIN implants than in NEO, MOR, and CON implants. Similarly, VH was significantly higher in RMO implants than in MOR and NEO ones. In addition, VH was higher in CON implants than in NEO ones. There were no significant differences in the proportions of titanium and aluminum in the mini-implant alloy of the five commercial brands. Conversely, the proportion of vanadium differed significantly between CON and MOR/NEO implants. Conclusions: Mini-implants of different brands presented distinct properties of hardness and composition of the alloy.
Keywords: orthodontics; dental materials; hardness.
Introduction
In recent decades, there has been a growing demand for orthodontic treatment in dental offices by adult patients, which required the development of an efficient and aesthetically-pleasing anchorage system to enable and expedite treatment. The orthodontic mini-implant is a temporary skeletal anchorage device that allows the orthodontist to work safely because it eliminates the ensuing side effects on teeth used as anchorage in conventional treatment and does not depend on patient compliance1. The ease of installation and removal, possibility of insertion in different intraoral regions, low cost, small healing time, and good patient acceptance all contribute to the diffusion of the technique2-4.
Several studies have demonstrated the clinical efficacy of this anchorage technique5-7, but clinical practice exposes some disadvantages such as screw breakage during installation, the possibility of osseointegration making removal difficult, and the lack of stability with subsequent mini-implant loss. Thus, further studies on the efficacy of mini-implants are needed to solve these issues.
The resistance of mini-implants is determined by the interaction between mini-implant design and chemical composition of the alloy. The alloy used for mini-implant production should be nontoxic, biocompatible, have good mechanical properties, and be stress, tension, and corrosion resistant8-9. The use of orthodontic mini-implants in different bone regions, including roots, requires that screws have reduced size and thickness and resist higher torsional loads than those required for insertion, in addition to orthodontic and orthopedic forces. To meet these requirements, the titanium alloy (Ti-6Al-4V) chosen for manufacture of mini-implants incorporates aluminum (Al) and vanadium (V) into its composition along with commercially pure titanium (cpTi)10. Ti-6Al-4V alloy has higher fatigue resistance than (cpTi), while having the same corrosion resistance and low toxicity11. The amount of each component in the alloy, in addition to the manufacturing quality, can alter its mechanical properties such as hardness and resiliency, which are responsible for the fracture resistance of the mini-implant.
Given the wide diversity of mini-implant types available in the market, this study aimed to evaluate the chemical composition and Vickers hardness (VH) of five orthodontic mini-implant commercial brands to determine whether their properties are suitable for clinical use.
Material and methods
Orthodontic mini-implants
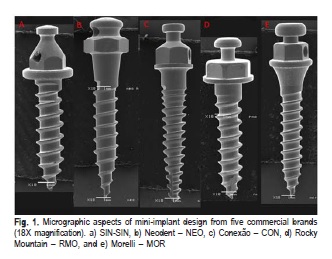
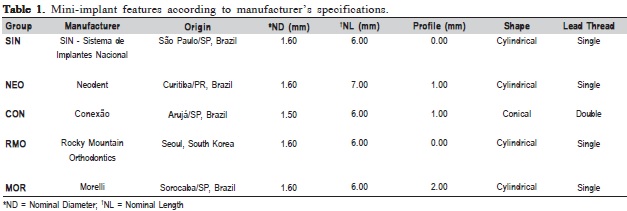
Thirty self-drilling mini-implants were used, six each from five commercial brands (SIN – SIN, Neodent – NEO, Conexão – CON, Rocky Mountain – RMO, and Morelli – MOR) with the largest number of similar characteristics to enable comparisons (Table 1). The macrostructural aspects of each mini-implant design are shown in Figure 1. Three mini-implants of each brand were used for the Vickers hardness analysis while the other three were used for the EDS analysis.
Vickers Hardness
Vickers hardness was determined by penetration length of the pyramidal diamond tip. A single operator previously calibrated by the repetition process performed hardness tests using a Shimadzu HMV-2micro hardness tester at a load of 300 gf and 4.904 N for 5 sec. The measurements were performed over the mini-implant head, which is the region with the highest stability for diamond penetration. Each of the three mini-implants from the five commercial brands was measured three times, totaling nine measurements per brand.
Energy dispersive X-ray spectroscopy (EDS)
The remaining mini-implants were used for semi quantitative analysis of alloy components. Each mini-implant was removed from its packaging only at the time of analysis so that the surface was not manipulated or contaminated by external agents, thus preserving the original characteristics. Six surface micrographs of each mini-implant (two of the mini-implant head, two of the body, and two closer to the mini-implant tip) were taken in panoramic view using a scanning electron microscope coupled with Noran Instruments EDS detector with a Vantage digital acquisition engine. The values for the chemical composition of the alloy were then obtained from these measurements.
Statistical Analysis
Due to the small number of samples, non-parametric tests were used for the statistical analysis using Bioestat 5.0 software (Belém, PA, Brazil). The Kruskall-Wallis complemented by the post-hoc test of Dunn was used to evaluate the statistical differences regarding the hardness and the chemical composition of the alloy between the brands. The Bioestat 5.0 software (Belém, PA, Brazil) was used for the analysis and the significance level was set at 5% (p<0.05) for all the tests.
Results
Vickers Hardness
There were significant differences in VH values between commercial brands. Vickers hardness was significantly higher in SIN implants than in NEO, MOR, and CON implants. Similarly, VH was significantly higher in RMO implants than in MOR and NEO ones. In addition, VH was higher in CON implants than in NEO ones (Table 2).

Energy dispersive X-ray spectroscopy (EDS)
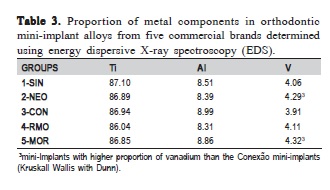
There were no significant differences in the proportions of titanium and aluminum in the mini-implant alloy of the five commercial brands. Conversely, the proportion of vanadium differed significantly between CON and MOR/ NEO implants. Even though the differences in mean values between brands were small, the low proportion of vanadium in the alloy causes small variations to be readily detectable (Table 3).
Discussion
Vickers hardness values showed little variation within each commercial brand except for CON implants. Conversely, VH values differed significantly between brands, and SIN and RMO implants exhibited the highest VH values.
Our findings are consistent with the study by Eliades et al. (2009)12, who evaluated mini-implants from a single commercial brand and found VH values of 342 ± 14 HV for the body and 354 ± 16 HV for the surface. These VH values are slightly higher than the ones found in our study, which ranged from 337 ± 4.66 HV (NEO) to 440 ± 35.14 HV (SIN). Moreover, the values of both studies are higher than the value (325.0 ± 10.1 HV) found by Lima et al. (2011)12, who evaluated implants made with the same alloy.
Energy dispersive X-ray spectroscopy (EDS) is a semi quantitative analysis that determines the chemical elements in the specimen and quantifies their approximate proportions. The results of our study confirmed the presence of Ti, Al, and V in mini-implants alloys, albeit at different proportions than those specified by manufacturers (Ti6Al4V). Nevertheless, the fact that we found a higher proportion of aluminum (approximately 8%) may be an artifact of the technique.
The difference in VH among brands may be due to differences in the proportion of titanium and vanadium in mini-implant alloys. Even though the proportion of titanium was similar in all commercial brands, titanium alloys can have different amounts of alpha-phase and beta-phase titanium, possibly due to differences in the manufacturing process of the alloy. For instance, Cotrim-Ferreira et al. (2010)11 showed that there were quantitative differences in alpha-phase and beta-phase titanium in mini-implant alloys from three commercial brands (SIN, Dewimed, and CON), even though they were within the guidelines of the "Technical Committee of European Titanium Producers" described in Publication ETTC-2.
The crystalline microstructure of the alloy, i.e., the amount of alpha and beta titanium in the alloy, is responsible for differences in its mechanical properties. An alloy with a higher amount of beta-phase titanium has higher tensile strength than an alloy with a higher amount of alpha-phase titanium, whereas alpha titanium has higher corrosion resistance than beta titanium10,14.
The addition of vanadium (a beta stabilizer) and an increase in temperature can both result in increased betaphase titanium in the alloy15. This study showed that there were differences in the proportion of vanadium among commercial brands. Even though these differences were not high, because the proportion of vanadium in the alloy is low, any small deviations are readily detectable and become statistically significant. Thus, the differences in VH among mini-implant brands may have been due to the proportion of vanadium in the alloy or to differences in heating and cooling temperatures during the manufacturing process of miniimplants14.
The difficulty in the casting process of titanium miniimplants may also have been responsible for the differences in VH observed in our study. The low density of titanium, its high melting temperature, and high chemical reactivity with surface elements and atmospheric gases make casting of this alloy extremely costly and laborious due to the need for special equipment to keep the titanium in a vacuum oxygen-free environment or in the presence of inert gases16-18. Additional tests such as X-ray diffraction are needed to test the hypothesis that titanium phases affect alloy hardness. The variation in alpha and beta phases of mini-implants may occur during manufacturing of the alloy or even during the manufacturing process of the mini-implant.
The knowledge of mini-implant hardness and composition is vital to assist in choosing the ideal commercial brand and model for each installation site, thus minimizing the risk of fracture. However, tensile strength of mini-implants is determined not only by their chemical composition and hardness, but mini-implant design is also fundamental for mini-implant choice since these parameters influence the possibility of the mini-implants fracture.
Based on the results of this research it can be concluded: 1. There were significant differences in Vickers hardness between SIN and Neodent/Morelli/Conexão; RMO and Neodent/Morelli; and Neodent and Conexão brands; 2. The proportion of vanadium in the alloy differed significantly between commercial brands; 3. The effect of these differences in the clinical practice needs to be test in clinical trials.
References
1. Kanomi R. Mini-implant for orthodontic anchorage. J Clin Orthod. 1997; 31: 763-7. [ Links ]
2. Nienkemper M, Pauls A, Ludwig B, Wilmes B, Drescher D. Multifunctional use of palatal mini-implants.J Clin Orthod. 2012; 46: 679-86.
3. Lehnen S, McDonald F, Bourauel C, Jäger A, Baxmann M. Expectations, acceptance and preferences of patients in treatment with orthodontic miniimplants: part II: implant removal. J Orofac Orthop. 2011; 72: 214-22.
4. Lehnen S, McDonald F, Bourauel C, Baxmann M. Patient expectations, acceptance and preferences in treatment with orthodontic mini-implants. A randomly controlled study. Part I: insertion techniques. J Orofac Orthop. 2011; 72: 93-102.
5. Rodriguez JC, Suarez F, Chan HL, Padial-Molina M, Wang HL. Implants for orthodontic anchorage: success rates and reasons of failures. Implant Dent. 2014; 23: 155-61.
6. Nienkemper M, Wilmes B, Pauls A, Drescher D. Multipurpose use of orthodontic mini-implants to achieve different treatment goals. J Orofac Orthop. 2012; 73: 467-76.
7. Motoyoshi M. Clinical indices for orthodontic mini-implants. J Oral Sci. 2011; 53: 407-12.
8. Pan CY, Chou ST, Tseng YC, Yang YH, Wu CY, Lan TH, et al. Influence of different implant materials on the primary stability of orthodontic miniimplants. Kaohsiung J Med Sci. 2012; 28: 673-8.
9. Serra G, Morais L, Elias CN, Semenova IP, Valiev R, Salimgareeva G, et al. Nanostructured severe plastic deformation processed titanium for orthodontic mini-implants. Mater Sci Eng C Mater Biol Appl. 2013; 33: 4197-202.
10. Morais LS, Serra GG, Muller CA, Andrade LR, Palermo EF, Elias CN, et al. Titanium alloy mini-implants for orthodontic anchorage: immediate loading and metal ion release. Acta Biomater. 2007; 3: 331-9.
11. Cotrim-Ferreira FA, Quaglio CL, Peralta RPV, Carvalho PEG, Siqueira DF. Metallographic analysis of the internal microstructure of orthodontic mini-implants. Braz Oral Res. 2010; 24: 438-42.
12. Eliades T, Zinelis S, Papadopoulos MA, Eliades G. Characterization of retrieved orthodontic miniscrew implants. Am J Orthod Dentofacial Orthop. 2009; 135: 10-7.
13. Lima GMA, Soares MS, Penha SS, Romano MM. Comparison of the fracture torque of different Brazilian mini-implants. Braz Oral Res. 2011; 25: 116-21.
14. AlSamak S, Bitsanis E, Makou M, Eliades G.Morphological and structural characteristics of orthodontic mini-implants. J Orofac Orthop. 2012; 73: 58-71.
15. Kent D, Wang G, Dargusch M. Effects of phase stability and processing on the mechanical properties of Ti-Nb based â Ti alloys. J Mech Behav Biomed Mater. 2013; 28: 15-25.
16. Donachie Jr MJ. Titanium. A Technical Guide. Ohio: ASM International; 2000.
17. Taira M, Moser JB, Greener EH. Studies of Ti alloys for dental castings. Dent Mater. 1989; 5: 45-50.
18. Voitik AJ. Titanium dental castings, cold worked titanium restorations-yes or no? Trends Tech Contemp Dent Lab. 1991; 8: 23-34.
 Correspondence:
Correspondence:
Nádia Lunardi
Av. Maria Antonia Camargo de Oliveira,170
CEP: 14707-120, Araraquara, SP, Brasil
E-mail: nadialunardi@yahoo.com.br
Received for publication: November 05, 2014
Accepted: January 29, 2015













